
Home

Research

Teaching

Publications

Contact info

|
|
Understanding the dynamics behind the photoisomerization of light-driven molecular rotary motors and switches
project financed by the EU
Nature has ingeniously designed molecular motors, which play a central role in biology as the means of movement in
living organisms. Chemistry is at an early stage of
building artificial molecular motors
at the bench. Currently, there is a high interest among the synthetic chemistry community in developing
molecular motors
and applying them in specific molecular devices (
functionalised surfaces,
nano-cars,
etc.).
Rational synthetic design of molecular motors, however, implies a thorough understanding of their functionality.
The thermochemical aspects of their operation are well understood, whereas their photo-dynamics still remains obscure and less amenable
to chemical modification. It is my primary objective to investigate the photochemistry of molecular light driven devices and by this to
fill the gap in our understanding of their mode of function. The outcome of this project will have far-reaching implications for
achieving a rational synthetic design of future molecular photo driven systems and will help to improve the principles used for
their design. This is a key to their application in future molecular devices in nanotechnology and synthetic biology.
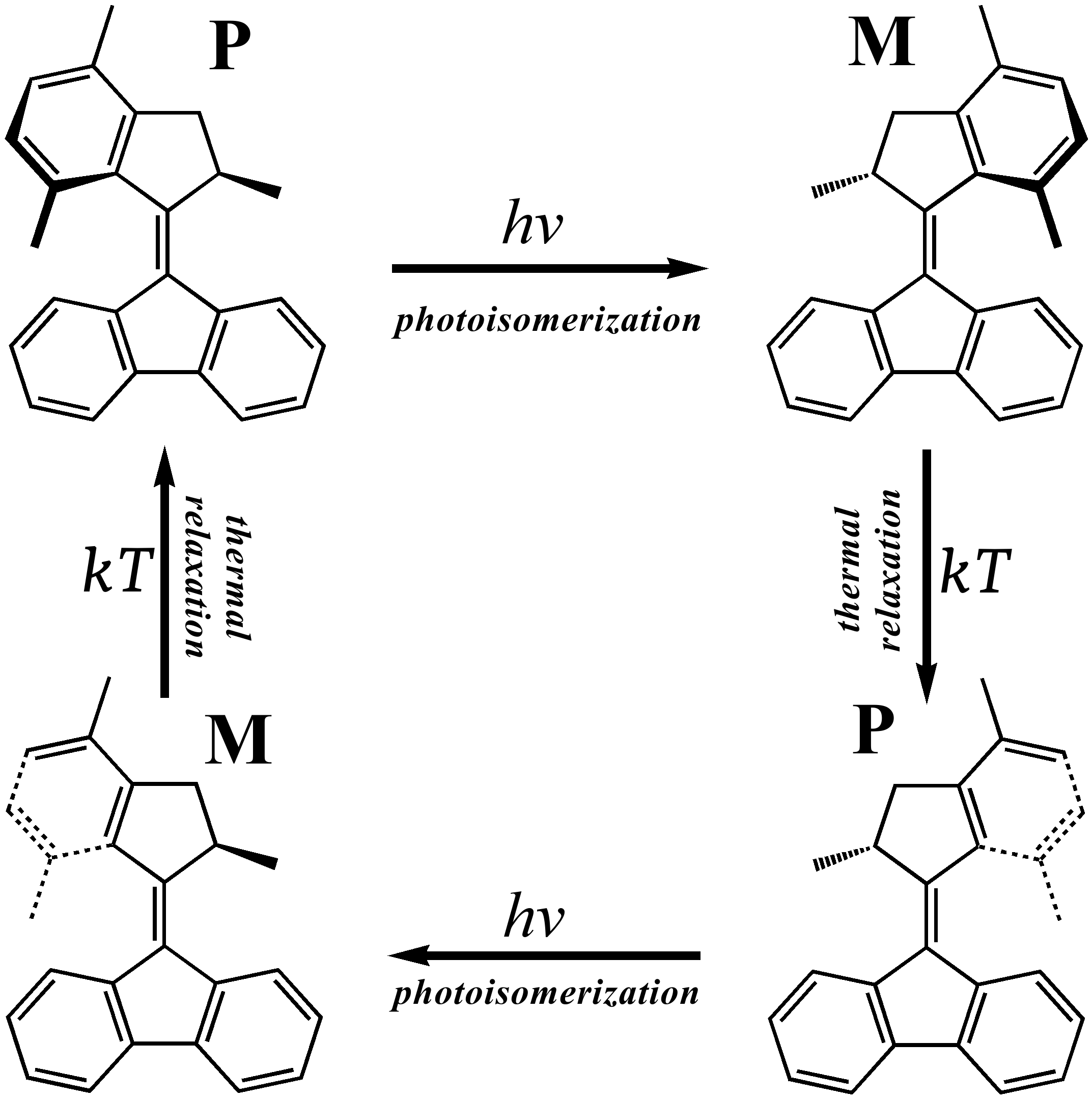
Working cycle of synthetic molecular motors
The
mode of action
of these motors is based on the periodic repetition of photo-isomerization and thermal relaxation steps,
which lead to a uni-directional rotation of one part of the molecule (rotor) with respect to another (stator).
Diagram to the right shows the sequence of steps that the motor molecule
undergoes
to achieve a full rotation cycle: The photoisomerization about the central double bond brings conformer with the P-helicity to the M-form,
which then undergoes a thermal helix inversion (M to P); after that the photoisomerization and helix inversion steps are repeated
to complete the loop. Photoisomerization step is the most important, as it leads to a large scale rotational motion (half-loop).
This step occurs on a time-scale of ca. 1.5 picosecond (ps), as suggested by both
theory
and
experiment. It takes
a few microseconds to complete the thermal helix inversion step.
|
|
The currently available synthetic motors display fairly low quantum efficiency (quantum yield of isomerization),
ca. 14%.
Roughly speaking only one out of seven light quanta results in rotation of the motor. Photoisomerization of the motor
occurs via special features on
the ground and excited electronic state
potential energy surfaces
known as
conical intersections.
The motor molecule in the scheme above reaches conical intersection at ca. 90° of twist about the central double bond
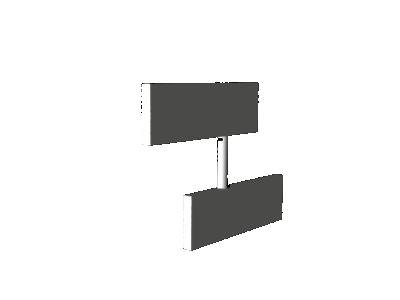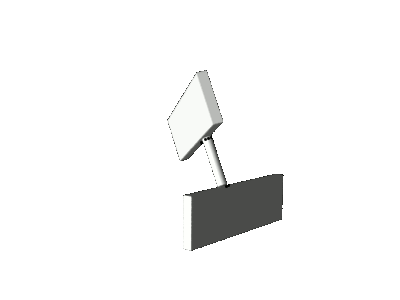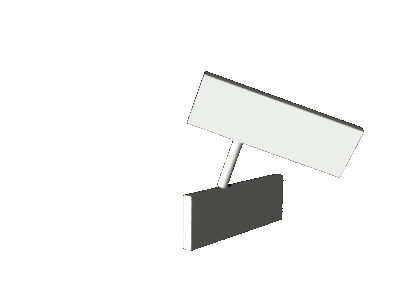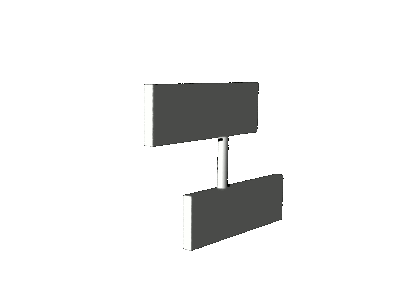
and an additional geometric distortion known as
pyramidalization. As a consequence of
pyramidalization, the motor molecule performs not a pure axial rotation about the central double bond, but a sort of precessional
motion as illustrated in the cartoons below.
precession
|
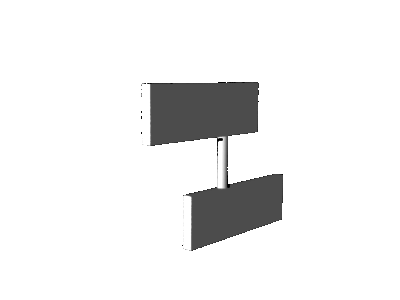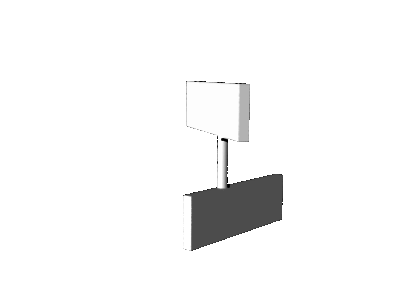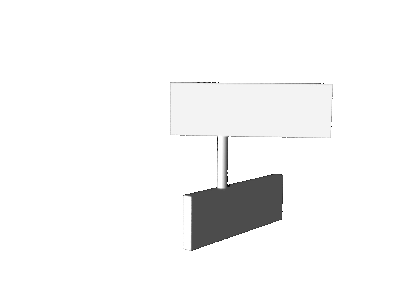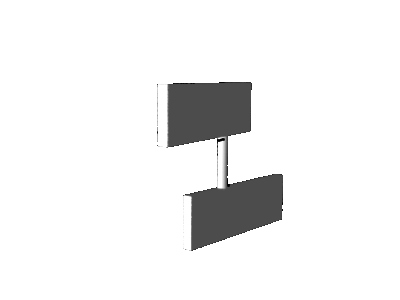
axial rotation
|
Furthermore, the need for pyramidalization leads to low accessibility of conical intersections and is the cause of the
low quantum efficiency of the existing molecular motors. It is therefore necessary to modify the motor molecule in
such a way that it could reach conical intersection without pyramidalization. In this way not only an axial rotation will
be achieved by the motor, but its quantum efficiency will be increased as a consequence of improved accessibility of conical
intersections.
|
What has been done so far
1. A new computational method for investigation of conical intersections in large molecules has been developed. For more information, see
publications 103
and 102.
2. The effect of strongly electron withdrawing substituents and heteroatoms on the geometry at which S0/S1
conical intersection is reached has been modelled computationally and it was established that introducing substituents such as
the cationic nitrogen removes the necessity to pyramidalize. Hence, a pure axial rotation can be achieved in molecular motors
shown in figures below.
3. A novel class of molecular motors based on
N-alkylated indanylidene pyrrolinium (NAIP) design
has been proposed. These motors are capable of performing a continuous axial rotation (at least in theory) and operating at low
temperatures. An example of a NAIP motor and its rotation cycle are shown below.
|
NAIP motor: the cartoon presents results of theoretical calculations
of all intermediate structures along the rotation pathway of the motor molecule.

|
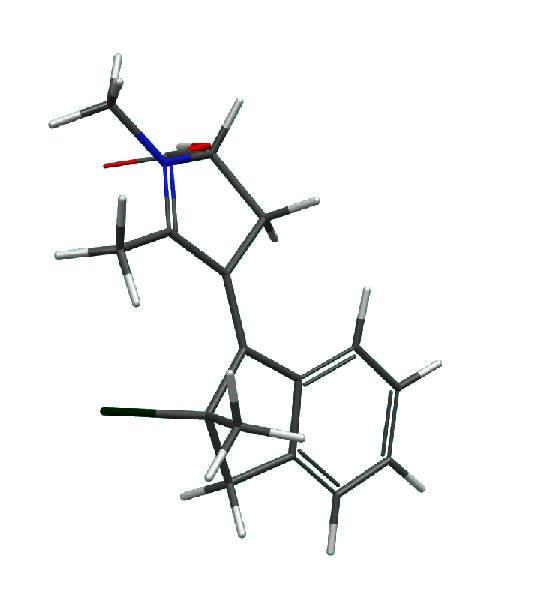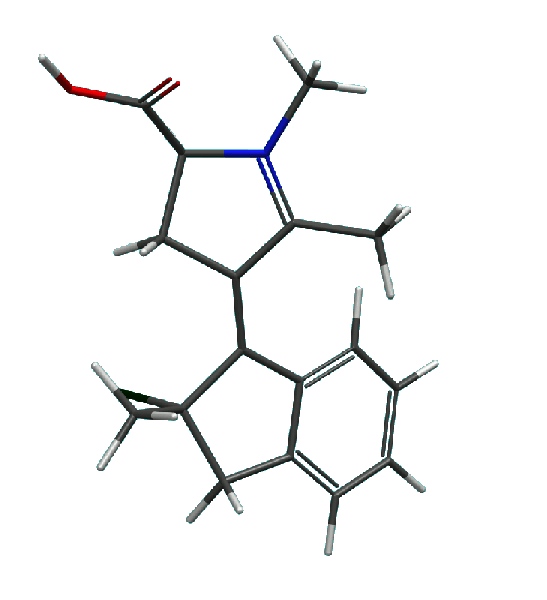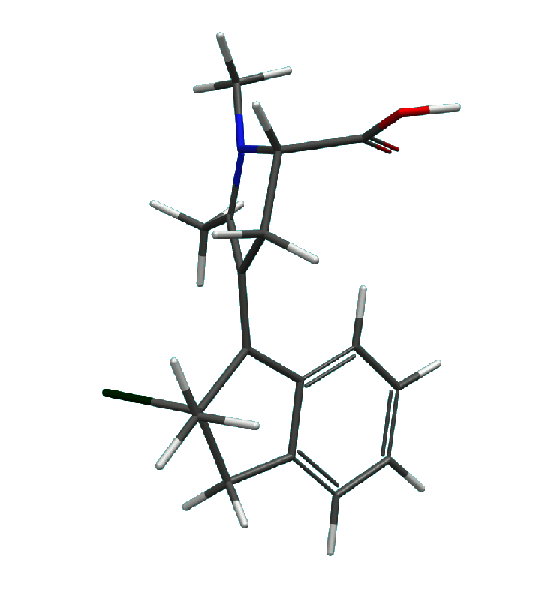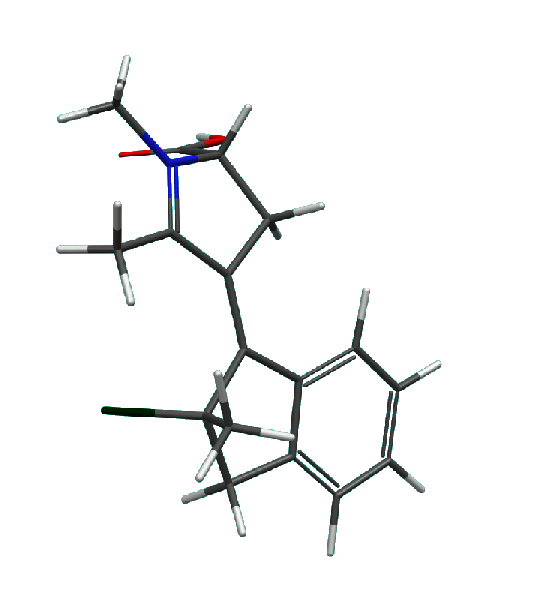
|
4. There is more to come...

For more information, see publications:
118,
111,
109,
103,
102,
99,
85,
79.
Top of the page
|
|